Metallurgy and Materials
SuperPro Designer is shipped with an expanding list of detailed examples that deal with extraction and purification of metals and rare earth elements, recycling of metals and manufacturing of advanced materials, such as those used in lithium-ion batteries. A brief description of the examples follows below. SuperPro’s databank includes physical and thermodynamic properties for more than 500 related materials. SuperPro Designer is the only process simulator that can handle modeling and optimization of batch and continuous metallurgical processes equally well. Batch processing is common in the manufacture and recycling of advanced and precious materials. Its integrated cost analysis capabilities facilitate estimation of capital as well as operating costs for new investments and the retrofitting and debottlenecking of existing manufacturing facilities. Its advanced environmental impact assessment capabilities facilitate waste minimization and ensure compliance with government regulations.
SchedulePro handles production planning and scheduling of integrated metal refineries as well as inventory tracking for input, intermediate and output materials. In addition, SchedulePro facilitates modeling, capacity analysis and debottlenecking of integrated metallurgical facilities that operate in batch and semi-continuous mode.
Hydrometallurgical Process Design and Economics with SuperPro Designer. This white paper covers the fundamentals of hydrometallurgical process synthesis, design, and economic evaluation. It also includes five detailed process examples modeled and analyzed with SuperPro Designer. You can test-drive all of the metallurgical examples of SuperPro Designer by downloading the trial edition of the tool from the downloads page of our website.
Manufacturing of Lithium-Ion Battery Cathode Material (NMC 811)

Lithium-ion batteries (LiBs) are high-capacity accumulators that find wide application in portable electronics and electric vehicles. The cathode of LiBs is made of a lithium transition metal oxide layered on aluminum foil whereas the anode consists of copperlayered carbon graphite. Due to the recent expansion of the electronics industry and upcoming switch to vehicle electrification, the production of LiBs is expected to reach 4,000,000 metric tons (MT) per year by 2025, corresponding to around 1,200,000 MT/year of cathode material.
This example analyzes a process for the generation of LiB cathode material from primary and secondary raw materials. The main product of the process is a Li-Ni-MnCo oxide with a Ni:Mn:Co ratio of 8:1:1, namely NMC 811. The raw materials used in the process include (i) older-generation NMC 111 scraps, (ii) metal Ni, and (iii) a mixture of Mn oxides. These raw materials are processed simultaneously in three parallel hydrometallurgical lines to generate the metal hydroxide precursors for NMC 811. The metal hydroxides are mixed with an equimolar amount of Li2CO3 and roasted under controlled air atmosphere to generate the final NMC 811 material.
The scale of the process analyzed in this example corresponds to production of 1.14 MT/hour of NMC 811, which results in annual throughput of 9,000 MT.
The NMC 811 process of this example runs in mixed batch-continuous mode. The hydrometallurgical lines and the mixing of raw materials operate in batch mode, whereas the thermal operations run in continuous mode.
The figure on the right displays the equipment occupancy chart (EOC) for eight consecutive batches of the sections that operate in batch mode. The equipment utilized in the process is listed on the y-axis and time on the x-axis. Each batch is represented by a unique color. The light-blue bars on the left of the chart represent the first batch. A new batch is initiated every 3 hours.
The NMC 111 scraps processing line utilizes two reactors (R-301 and R-301b) operating in staggered mode in order to reduce the cycle time of the overall process to 3 hours. The EOC in SuperPro Designer facilitates cycle time reduction and debottlenecking studies.


Demand for labor, utilities, materials and other resources varies with time in batch processing. The chart on the left displays labor demand as a function of time for 12 consecutive batches of the NMC 111 manufacturing process. Labor demand ranges from 10 to 23 operators during a typical shift. This information is useful for estimating the labor requirement and associated labor cost for batch manufacturing facilities.
Small changes in the schedule can reduce the maximum number of operators from 23 to 20 and lower the labor cost. However, the changes may have a negative impact on the cycle time and the overall throughput of the process. Such trade-offs can be readily analyzed and evaluated with SuperPro Designer.
Charts displaying utility requirements as a function of time are useful for the sizing of utility systems (e.g., generation & supply of power, steam, purified water, etc.) for batch manufacturing facilities.
Recycling of Lithium-Ion Batteries
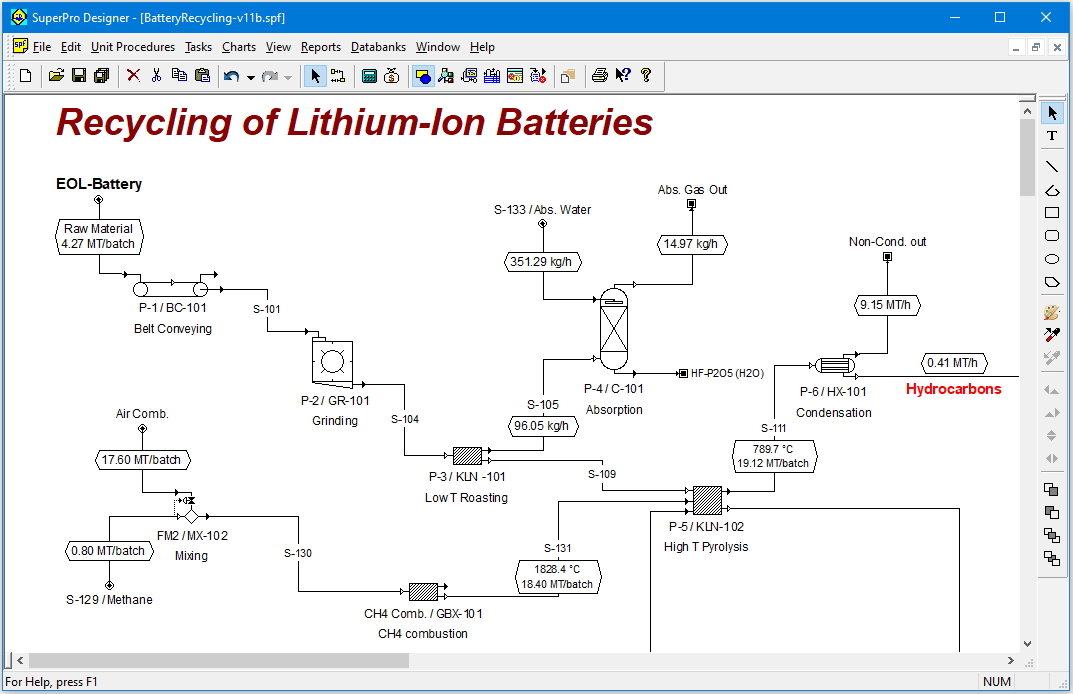
This example analyzes the recycling of Lithium-ion batteries (LiBs) for portable electronics. Recycling of batteries is highly beneficial to society because it reduces the depletion of natural resources and minimizes environmental impact.
The process of this example begins with a physical pretreatment section to free the electrode powder and separate other valuable components. That is followed by a hydrometallurgical line to extract, purify and recover the metals contained in the electrode powder. The hydrometallurgical line is based on leaching, primary purification by precipitation, secondary purification by solvent extraction with D2EHPA and CYANEX 272, and metals recovery by crystallization / precipitation.The process of this example enables the recycling of 17,000 metric tons (MT) of LiBs per year and generates 1,700 MT/year of Li2CO3, 1,750 MT/year of NiCO3, 2,400 MT/year of MnSO4 and 2,350 MT/year of CoSO4.
The process of this example runs in a mixed batch-continuous mode. The physical pretreatment operations and the crystallization units run in continuous mode whereas leaching, precipitations and solvent extraction run in batch mode.
The figure on the right displays the equipment occupancy chart (EOC) for eight consecutive batches. The equipment utilized in the process is listed on the y-axis and time on the x-axis. Each batch is represented by a unique color.
This example analyzes two cases (A and B) that differ in cycle times. Case B has access to two additional reactors that were allocated to the leaching and nickel dissolution steps which were the bottlenecks in case A.
The availability of two extra reactors operating in staggered mode for the bottleneck steps reduced the cycle time of the process from 3.25 to 2 hours and increased the throughput of the overall process by more than 60%. Cycle time reduction and debottlenecking studies are common applications of SuperPro Designer.

Extraction of Lithium from Spodumene Ore
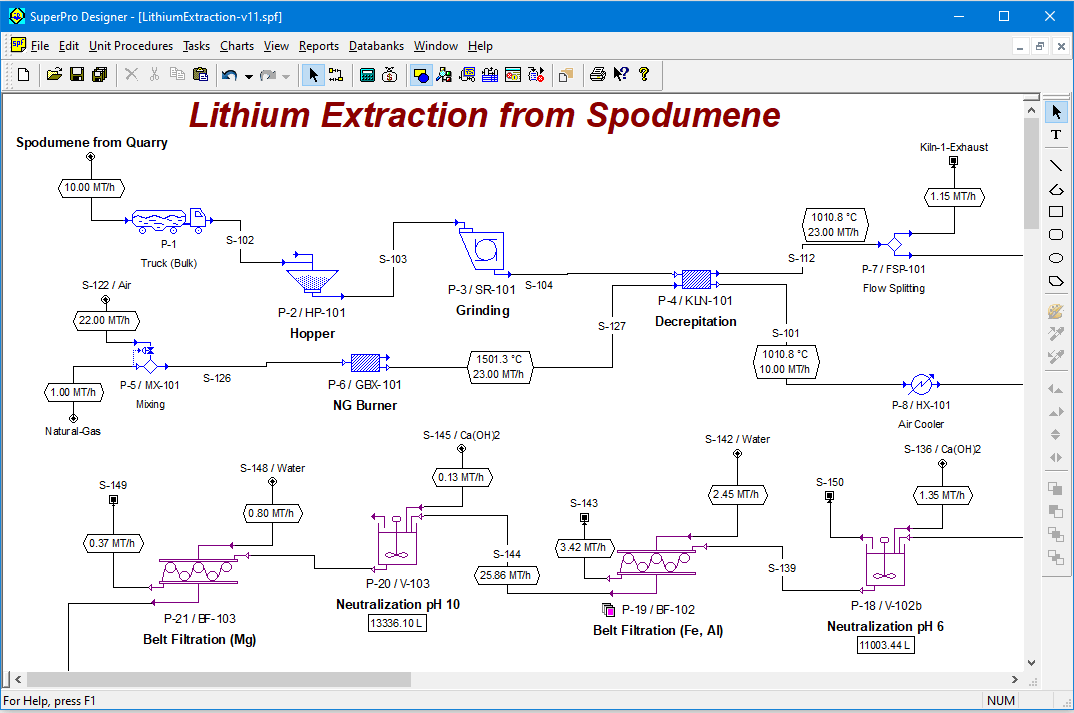
Spodumene (LiAlO6Si2) is the most common mineral processed commercially to produce lithium compounds. The world production of lithium via spodumene is around 80,000 metric tons (MT) per year, primarily from the Greenbushes pegmatite of Western Australia and secondarily from Chinese and Chilean sources.
This example analyzes a continuous process that generates 1.3 MT/h of batterygrade Li2CO3 (10,460 MT/year) from 10 MT/h (79,200 MT/year) of spodumene concentrate containing 75-87% spodumene along with impurities such as quartz (SiO2), alumina (Al2O3), annite (KFe3AlSi3O10(OH)2) and phlogopite (KMg3AlSi3O10(OH)2). The flowsheet is divided into three ections: Decrepitation & Sulfation, Leaching & Neutralization, and Product urification.
In Decrepitation & Sulfation, most minerals contained in the concentrate are transformed to their respective sulfates upon converting the less reactive αspodumene to β-spodumene. In Leaching & Neutralization, the metal sulfates are dissolved in water prior to purification of the obtained solution by step-wise increase of the solution pH up to 11. Finally, the Li2CO3 is obtained by carbonation of the lithium sulfate solution and crystallization.
A variation of this process has been commercialized by Talison Lithium in Australia and Sichuan Tianqi Lithium Industries in China.
Rare Earth Elements (REE) Production
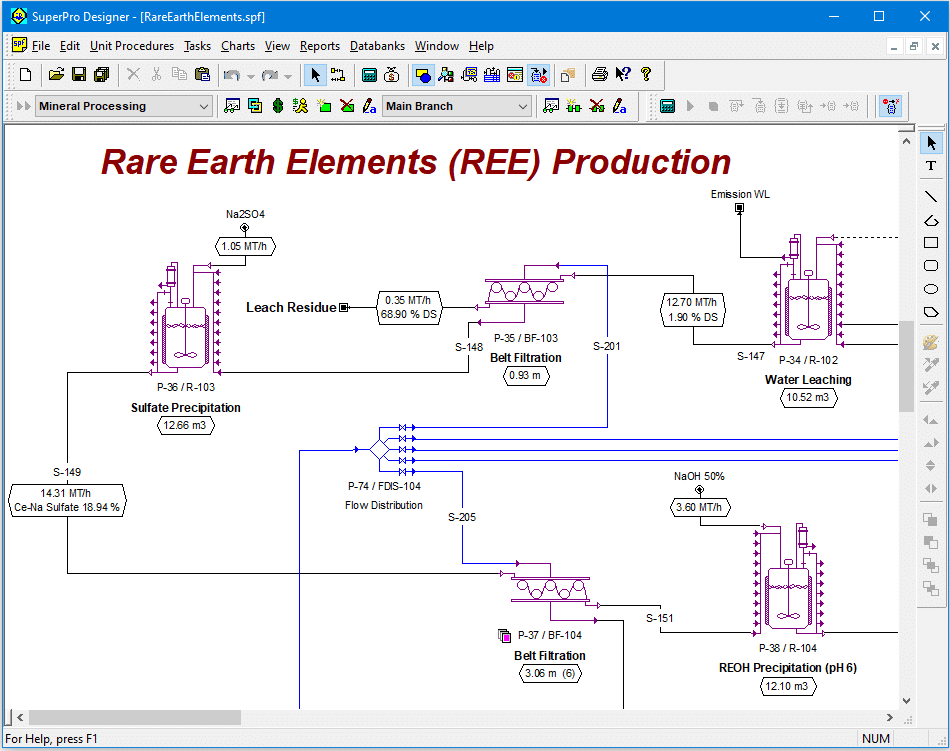
Rare earth elements (REEs) include the 15 elements of the lanthanide group from lanthanum to lutetium and are usually split into two sub-groups, namely light rare earth elements (LREEs) and heavy rare earth elements (HREEs). REEs play important roles in electronics, batteries, high-power magnets, catalysis, ceramics and other fields. REEs cannot be substituted by other elements and due to the limited availability of rare earth resources, REEs are considered essential materials for the industrial development of a country.
This example analyzes the production of high-quality rare earth oxides (REOs) and other marketable products from a fluorocarbonate iron-niobium ore containing 4% of light REOs. The process handles 50 metric tons (MT) of raw ore per hour (396,000 MT/year) and generates 1.25 MT/h of cerium dioxide (CeO2), 0.62 MT/h of lanthanum oxide (La2O3) and 0.55 MT/h of a mixture of neodymium and praseodymium oxides (NdO3-Pr6O11) as well as other marketable co-products such as magnetite, monazite, iron-niobium, and fluorite concentrates.
The analyzed process features a mineral processing section based on comminution, magnetic separation and flotation and a hydrometallurgical section for the extraction and recovery of the LREEs. In the mineral processing section, the REO concentration in the ore is increased from the initial 4% to 70% via separation and recovery of the co-products. In the hydrometallurgical section, the LREEs are extracted from the concentrate into sulfuric acid by sulfatation-roasting, separated from other impurities trough a sequence of precipitation-dissolution, separated from each other by solvent extraction, and recovered as oxides via precipitation-calcination.
Extraction of Zirconium, Uranium, Hafnium and Silica Gel from Zircon
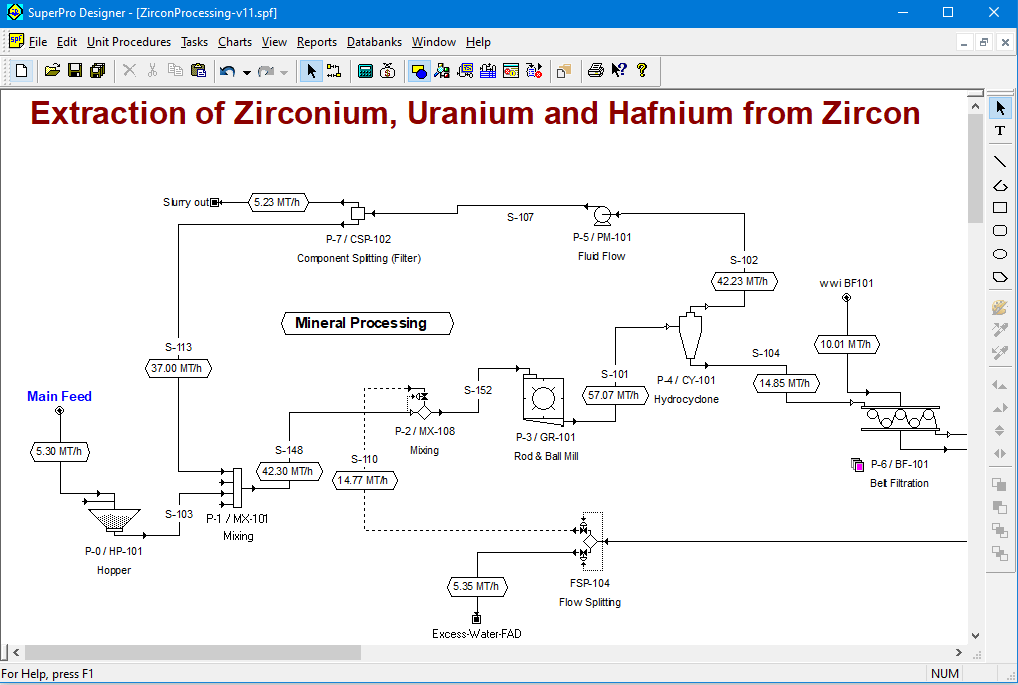
Approximately 1.4 million metric tons (MT) of zirconium are produced every year to be used as zirconia (ZrO2) in ceramics, refractory materials, glazes, foundry mold and abrasive grits. One of the main natural sources of zirconium is the zircon mineral (ZrSiO4), also known as zircon sand. This mineral is abundant in Australia, although China, South Africa, USA and India play significant roles in its mining as well. Since these minerals are often accompanied by uranium and hafnium species, the production of zirconium is often associated with the generation of hafnium and uranium by-products.
This example analyzes a process that produces around 2.3 MT/hour of commercial grade zirconia from zircon ore. The by-products of the process include silica, uranium dioxide and a Hf-rich stream. The process starts with the mineral processing of the crude ore followed by hydrometallurgical operations. The mineral processing concentrates the zircon minerals by removing the clay and hard rocks contained in the ore. The hydrometallurgical section features alkali-fusion, leaching, sulfation and primary purification prior to solvent extraction. The solvent extraction is performed with trioctylamine (TOA) in a counter-current circuit of 4 multi-stage mixer-settlers.
Copper-Nickel (Cu-Ni) Matte Leaching

Copper-nickel matte is a mixture of metal sulfides generated during the pyrometallurgical smelting of copper. Although the exact composition of this material depends on the upstream raw materials and furnace conditions, smelting polymetallic minerals results in a matte that is typically rich in copper minerals such as chalcocite (Cu2S) and bornite (Cu5FeS4) but also nickel as heazlewoodite (Ni3S2), iron as pyrite (FeS2) and other metal sulfide impurities.
This example analyzes a hydrometallurgical process for the treatment of coppernickel (Cu-Ni) matte. The process features high pressure leaching-metathesis and downstream operations. Pressure leaching dissolves a controlled amount of copper as sulfate, which in turn induces metathesis reactions with other metal sulfides. This results in the selective dissolution of nickel with accumulation of Cu sulfides in the solid residue. Nickel is recovered by precipitation as carbonate (NiCO3) from the pregnant leach solution after a primary crystallization, redissolution of the crystals in water and iron removal by precipitation as goethite (FeOOH). The process model of this example includes a centralized water recirculation system that minimizes the demand for fresh water and the generation of wastewater.
The process runs in continuous mode with an inlet flowrate of 4 metric tons (MT) of matte per hour (31,680 MT/year) and generates 1.23 MT/hour of NiCO3 (9,730 MT/year). An enriched copper concentrate fraction with a flowrate of 2.71 MT/hour (21,427 MT/year) is generated as a co-product of the process.
Printed Circuit Boards (PCBs) Recycling
The global production of electronic waste reached 54 million metric tons (MT) in 2019 and is projected to rise to 75 million MT by 2030. As key components in all electric and electronic devices, printed circuit boards (PCBs) represent 3-6% of the total amount of waste of electric and electronic equipment (WEEE). Yet, due to the presence of valuable metals such as copper, silver and gold, they are associated with over 40% of the total revenues from WEEE recycling. This example analyzes a facility that processes 15,000 MT/year of PCBs. The process runs continuously and features mechanical pretreatment and multiple hydrometallurgical sections to separate and recover 2,586 MT/year of electrolytic copper, 1,680 MT/year of aluminum hydroxide, 765 MT/year of zinc hydroxide, 13.48 MT/year of silver and 1.31 MT/year of gold among other materials.
Comminution frees the metal values and enables the magnetic separation of ferrous metals. An acidic-oxidizing leaching of the non-ferrous fraction in sulfuric acid / hydrogen peroxide extracts copper, which is later recovered via electrowinning after precipitation & recovery of the co-extracted aluminum as hydroxide. Part of the spent electrolyte is discharged as a wastewater prior to cementation of the residual copper, precipitation of zinc as hydroxide, and crystallization of the sodium sulfate accumulated throughout neutralization operations. Silver is recovered from the copper leach residue via leaching in nitric acid and electrowinning. Gold is leached out of the silver leach residue using hydrochloric acid / hydrogen peroxide and recovered via reduction with ferrous chloride.

End-of-Life Solar Photovoltaic Panels Recycling

Photovoltaic (PV) panels are being widely deployed worldwide to generate electricity from sun light. The installed capacity in 2020 was around 300 GW, and it is expected to reach 4,500 GW by 2050. The average life of PV panels is around 25 years. The current need for end-of-life PV panel recycling is around 200,000 metric tons per year (MT/year). The recycling demand is expected to reach 80 million MT/year by 2050.
This SuperPro Designer example analyzes a physico-chemical process for recycling of end-of-life PV panels. The process involves dry pre-treatment, wet chemical treatment, and hydrometallurgical processing. The dry pre-treatment begins with the manual dismantling of the aluminium frames and proceeds with comminution and sieving. The chemical treatment is based on the alteration of the ethylene vinyl acetate (EVA) used as encapsulating agent by means of cyclohexane, thus resulting in the separation of Tedlar, glass, metal contacts and EVA sheets. Glass and metal particles are separated and recovered by air classification. The silver and silicon encapsulated within the EVA layers are recovered by hydrometallurgical operations upon thermal decomposition of EVA. The overall recovery of materials is more than 90%.
The facility processes 10 MT/hour of PV panels, corresponding to 79,200 MT/year. It enables the recovery of 7,920 MT/year of aluminum frames, 35,500 MT/year of coarse glass particles, and 33 MT/year of silver along with additional valuable materials.
Copper and Gold Bioleaching
Copper and gold are two important metal commodities that find wide application across many industrial and non-industrial sectors. Due to the decreasing availability of gold deposits with a simple mineralogy, a large proportion of gold is expected to be produced from complex ores containing significant amounts of copper sulfide minerals. In these ores, the presence of native gold locked within other metal sulfides does not allow the use of conventional technologies based on cyanide leaching. A more effective strategy to recover gold from these ores, which are also known as refractory, involves the preliminary bacterial leaching to oxidize the sulfide minerals and dissolve copper, thereby enhancing the accessibility to the locked gold.
This SuperPro example analyzes a bio-hydrometallurgical process to extract and recover copper and gold from a Cu-Au refractory concentrate. The designed flowsheet is based on the preliminary bioleaching of the concentrate to dissolve the metal sulphides while leaving gold in the solid residue. Copper is recovered from the bioleach pregnant solution by electrowinning, after purification by solvent extraction. Gold is extracted from the bioleach residue by thiosulfate-ammonia and recovered from the pregnant leach solution by electrowinning upon purification by ion-exchange chromatography.
The manufacturing plant analyzed in this example treats 328,000 metric tons (MT) of concentrate per year and generates 87,000 MT of copper and 3.65 MT of gold.

Aluminum Production
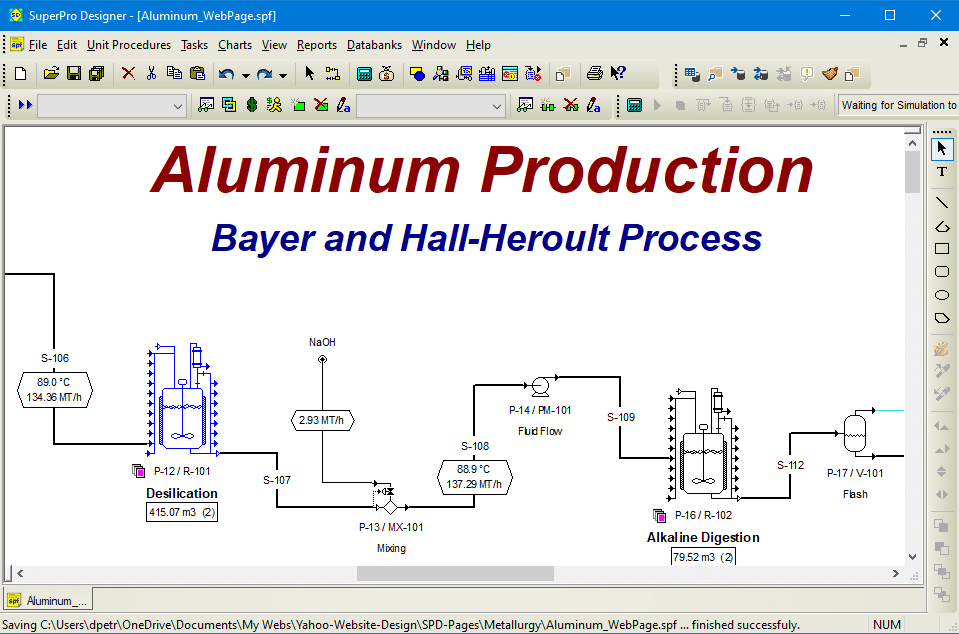
Aluminum is a commodity non-ferrous metal that is widely used in construction, transportation, packaging, and consumer equipment due to its lightness, high thermal and electrical conductivity, corrosion resistance, and workability. Around 70% of the over 65 million metric tons (MT) of aluminum produced worldwide is generated from primary resources such as bauxite ore. This is done via the Bayer and Hall-Heroult processes. The Bayer process accomplishes the extraction and separation of aluminum from bauxite by alkaline pressure leaching, precipitation of aluminum hydroxide and calcination of the hydroxide to alumina. The Hall-Heroult process reduces the alumina to aluminum via electrowinning from molten salt medium.
This SuperPro example analyses a metallurgical facility that utilizes the Bayer and Hall-Heroult processes to produce aluminum from bauxite ore. The facility treats 425,000 MT of bauxite ore per year and generates 85,000 MT/year of aluminum.


